Nernst equation
 Generated by Llama 3.3-70B
Generated by Llama 3.3-70BExpansion Funnel Raw 83 → Dedup 24 → NER 12 → Enqueued 0
| Nernst equation | |
|---|---|
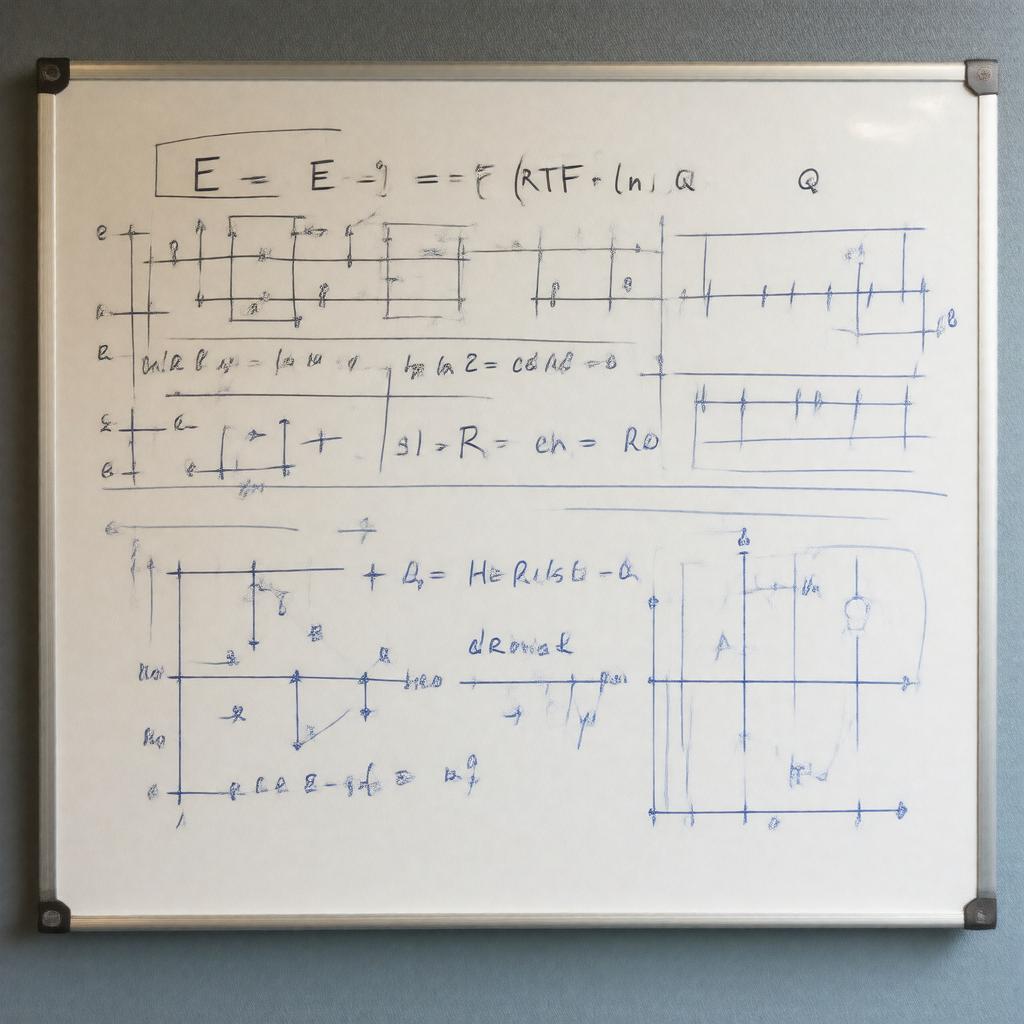 | |
| Name | Nernst equation |
| Equation | E = E° - (RT/nF) \* ln(Q) |
Nernst equation. The Nernst equation is a fundamental concept in electrochemistry, developed by Walther Nernst, a Nobel Prize in Chemistry laureate, in collaboration with Max Planck and Svante Arrhenius. This equation relates the electrode potential of a cell to the standard electrode potential and the concentrations of the chemical species involved, as described by Le Chatelier's principle and the work of Josiah Willard Gibbs. The Nernst equation has been widely used in various fields, including chemistry, physics, and materials science, with contributions from notable scientists such as Marie Curie, Ernest Rutherford, and Niels Bohr.
Overview
The Nernst equation is a mathematical expression that describes the relationship between the electrode potential of a cell and the concentrations of the chemical species involved, as studied by Heinrich Hertz and James Clerk Maxwell. It is commonly used to calculate the potential of an electrochemical cell, such as a galvanic cell or an electrolytic cell, under non-standard conditions, as described by Hermann von Helmholtz and Ludwig Boltzmann. The equation takes into account the standard electrode potential of the cell, the temperature of the system, and the concentrations of the reactants and products, as investigated by Wilhelm Ostwald and Jacobus Henricus van 't Hoff. The Nernst equation has been applied in various fields, including corrosion science, bioelectrochemistry, and energy storage, with contributions from researchers at MIT, Stanford University, and the University of Cambridge.
Derivation
The Nernst equation can be derived from the Gibbs free energy equation, which relates the energy of a system to its entropy and enthalpy, as described by Rudolf Clausius and Lars Onsager. The derivation involves the use of thermodynamic equations, such as the Nernst-Planck equation, and the assumption of equilibrium conditions, as studied by Ilya Prigogine and Nikolay Semyonov. The resulting equation is a logarithmic expression that relates the electrode potential to the concentrations of the chemical species involved, as investigated by Manfred Eigen and Ronald Norrish. The Nernst equation has been used to study various electrochemical phenomena, including electrode kinetics and mass transport, with contributions from researchers at Caltech, Harvard University, and the University of Oxford.
Applications
The Nernst equation has a wide range of applications in various fields, including electrochemistry, materials science, and biomedical engineering, with contributions from researchers at NASA, IBM, and the European Organization for Nuclear Research. It is commonly used to design and optimize electrochemical cells, such as batteries and fuel cells, as studied by Thomas Edison and Nikola Tesla. The equation is also used to study corrosion phenomena and to develop corrosion protection strategies, as investigated by Ulf Wiedensohler and Andrei Geim. Additionally, the Nernst equation has been used in biomedical applications, such as the development of biosensors and implantable devices, with contributions from researchers at Johns Hopkins University and the University of California, Berkeley.
Limitations and extensions
While the Nernst equation is a powerful tool for understanding electrochemical phenomena, it has several limitations and extensions, as discussed by Stephen Hawking and Brian Greene. One of the main limitations is that the equation assumes equilibrium conditions, which may not always be the case in real-world systems, as studied by Ilya Prigogine and Nikolay Semyonov. Additionally, the equation does not take into account non-ideal behavior of the electrolyte, such as ion pairing and electrolyte interactions, as investigated by Manfred Eigen and Ronald Norrish. To address these limitations, various extensions of the Nernst equation have been developed, including the Butler-Volmer equation and the Marcus theory, with contributions from researchers at MIT, Stanford University, and the University of Cambridge.
Example calculations
The Nernst equation can be used to calculate the electrode potential of a cell under various conditions, as demonstrated by Richard Feynman and Murray Gell-Mann. For example, the equation can be used to calculate the potential of a hydrogen electrode in an acidic solution, as studied by Heinrich Hertz and James Clerk Maxwell. The calculation involves plugging in the values of the standard electrode potential, the temperature, and the concentrations of the reactants and products, as described by Wilhelm Ostwald and Jacobus Henricus van 't Hoff. The resulting potential can then be used to predict the behavior of the cell under various conditions, such as current density and pH, with contributions from researchers at Caltech, Harvard University, and the University of Oxford. The Nernst equation has been applied in various fields, including chemistry, physics, and materials science, with notable contributions from Marie Curie, Ernest Rutherford, and Niels Bohr, and institutions such as the National Institutes of Health, the European Space Agency, and the Chinese Academy of Sciences. Category:Electrochemistry