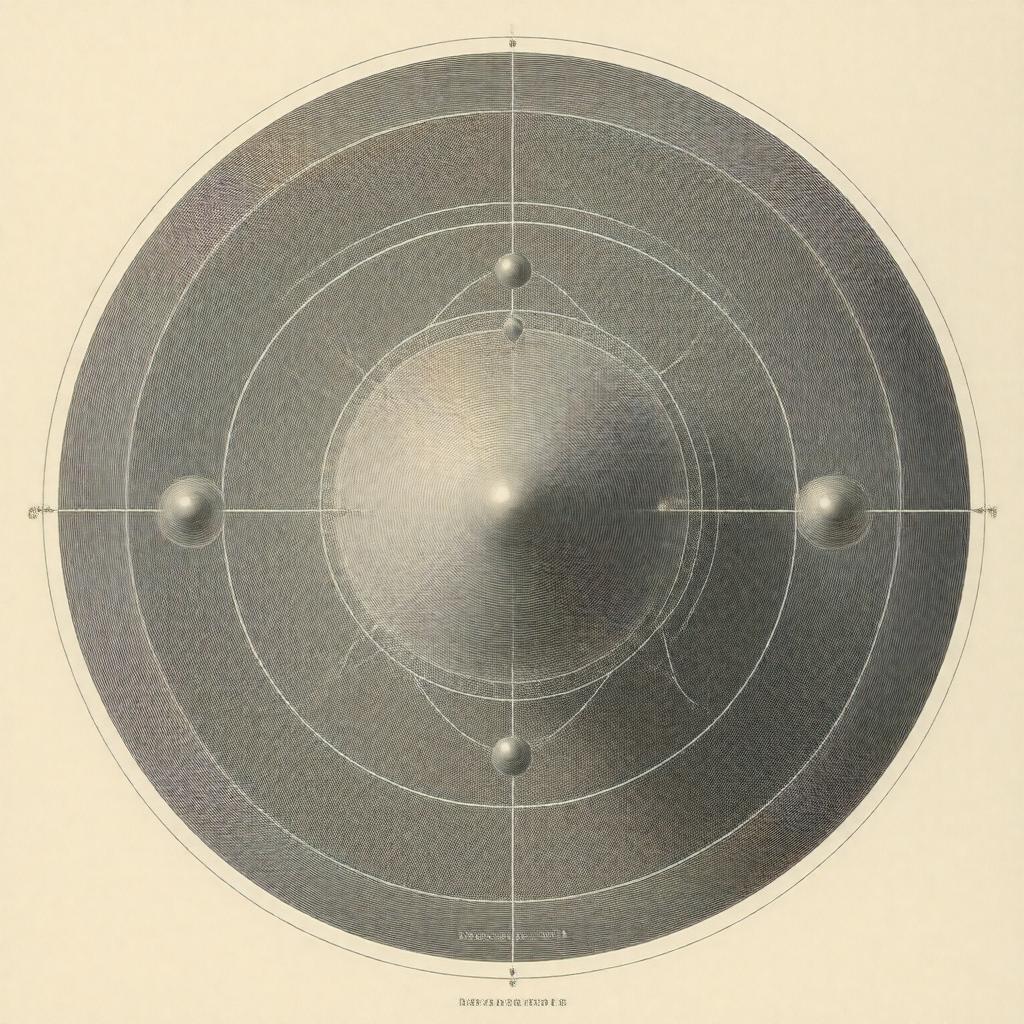
Rutherford model of the atom
 Generated by GPT-5-mini
Generated by GPT-5-miniExpansion Funnel Raw 57 → Dedup 0 → NER 0 → Enqueued 0
| Rutherford model of the atom | |
|---|---|
 | |
| Name | Rutherford model of the atom |
| Author | Ernest Rutherford |
| Year | 1911 |
| Field | Atomic physics |
| Predecessor | Plum pudding model |
| Successor | Bohr model |
Rutherford model of the atom The Rutherford model of the atom, proposed by Ernest Rutherford in 1911, recharacterized atomic structure by introducing a compact, positively charged nucleus surrounded by light, negatively charged particles. It superseded the Plum pudding model associated with J. J. Thomson and set the stage for later quantum and nuclear theories developed by figures such as Niels Bohr, Arnold Sommerfeld, Werner Heisenberg, and Erwin Schrödinger. The model arose from experimental work performed by collaborators including Hans Geiger and Ernest Marsden at institutions like the University of Manchester and influenced research at laboratories including the Cavendish Laboratory and the Royal Institution.
Background and development
Rutherford's proposal followed investigations into radioactivity by Henri Becquerel, Marie Curie, and Pierre Curie, and guidance from mentors such as J. J. Thomson and contemporaries including Rutherford's students at McGill University. Developments in experimental apparatus at universities like Trinity College, Cambridge and facilities such as the Kaiser Wilhelm Society enabled precise scattering measurements. The intellectual climate included debates over atomic structure involving figures like John Dalton (historical atomic theory), Dmitri Mendeleev (periodic system implications), and theoretical critiques from physicists in centers such as the University of Göttingen and the University of Zurich.
Experimental basis (gold foil experiment)
The empirical foundation was the gold foil experiment, in which alpha particles emitted by sources including Radium samples were directed at thin metal foils; key personnel included Hans Geiger and Ernest Marsden working under Rutherford. Detection techniques drew on scintillation methods refined in laboratories like the Cavendish Laboratory and instrumentation influenced by designers at places such as the Royal Society's facilities. Unexpected large-angle scattering events contradicted predictions from models advocated by J. J. Thomson and prompted Rutherford to posit a dense central nucleus, a conclusion that reverberated through communities at institutions such as Imperial College London and the University of Oxford.
Key features and assumptions
Rutherford asserted that most atomic mass and positive charge concentrate in a small nucleus containing protons, a notion resonant with contemporary work by Eugene Goldstein and later extended by James Chadwick's discovery of the neutron. Electrons orbit this nucleus much like planetary bodies, an analogy borrowed from discussions among scientists at the Royal Society and educators at institutions like King's College London. The model assumed classical mechanics and Coulomb interactions described by laws developed by Isaac Newton and Charles-Augustin de Coulomb, without quantized energy levels that would later be introduced by Niels Bohr and formalized in quantum theory by Max Planck and Albert Einstein.
Successes and limitations
Rutherford's model explained observations that the Plum pudding model could not, such as large-angle alpha scattering and atomic mass concentration, winning support from peers at meetings of the British Association for the Advancement of Science and citations in work by researchers at the Institut Henri Poincaré. However, it failed to account for atomic stability and discrete spectral lines measured by spectroscopists like Johann Balmer and interpreted by Niels Bohr; classical electrodynamics from contributors like James Clerk Maxwell predicted radiative collapse of orbiting electrons, a problem highlighted in critiques by theorists at the University of Copenhagen and the Sorbonne.
Influence on later atomic models
Rutherford's nucleus concept directly inspired Niels Bohr's model, which incorporated quantum postulates and explained hydrogen spectra investigated by spectroscopists at institutions such as Uppsala University and Leiden University. The nuclear paradigm guided nuclear physics research conducted by collaborators and successors including Ernest Walton, John Cockcroft, and Enrico Fermi at laboratories including the Cavendish Laboratory, University of Cambridge, and the University of Rome. Rutherford's ideas also framed discoveries in particle physics by teams at the CERN precursor institutions and encouraged explorations leading to the identification of subatomic constituents by groups at the Lawrence Berkeley National Laboratory and the Fermi National Accelerator Laboratory.
Experimental confirmations and refinements
Subsequent experiments—alpha scattering studies by investigators such as Geiger–Marsden follow-ups, mass spectrometry advances by Francis Aston, and later neutron detection by James Chadwick—confirmed and refined the nuclear picture. The emergence of quantum mechanics through work by Werner Heisenberg, Erwin Schrödinger, Paul Dirac, and experimental verifications at institutions like the Max Planck Institute resolved the stability and spectral issues. High-energy scattering experiments at facilities including Cavendish Laboratory successors, Brookhaven National Laboratory, and CERN probed nuclear structure further, culminating in models of nucleon composition informed by discoveries from groups led by figures such as Murray Gell-Mann and George Zweig.
Category:Atomic models