Atomic Physics
 Generated by GPT-5-mini
Generated by GPT-5-miniExpansion Funnel Raw 82 → Dedup 5 → NER 5 → Enqueued 3
| Atomic Physics | |
|---|---|
 | |
| Name | Atomic Physics |
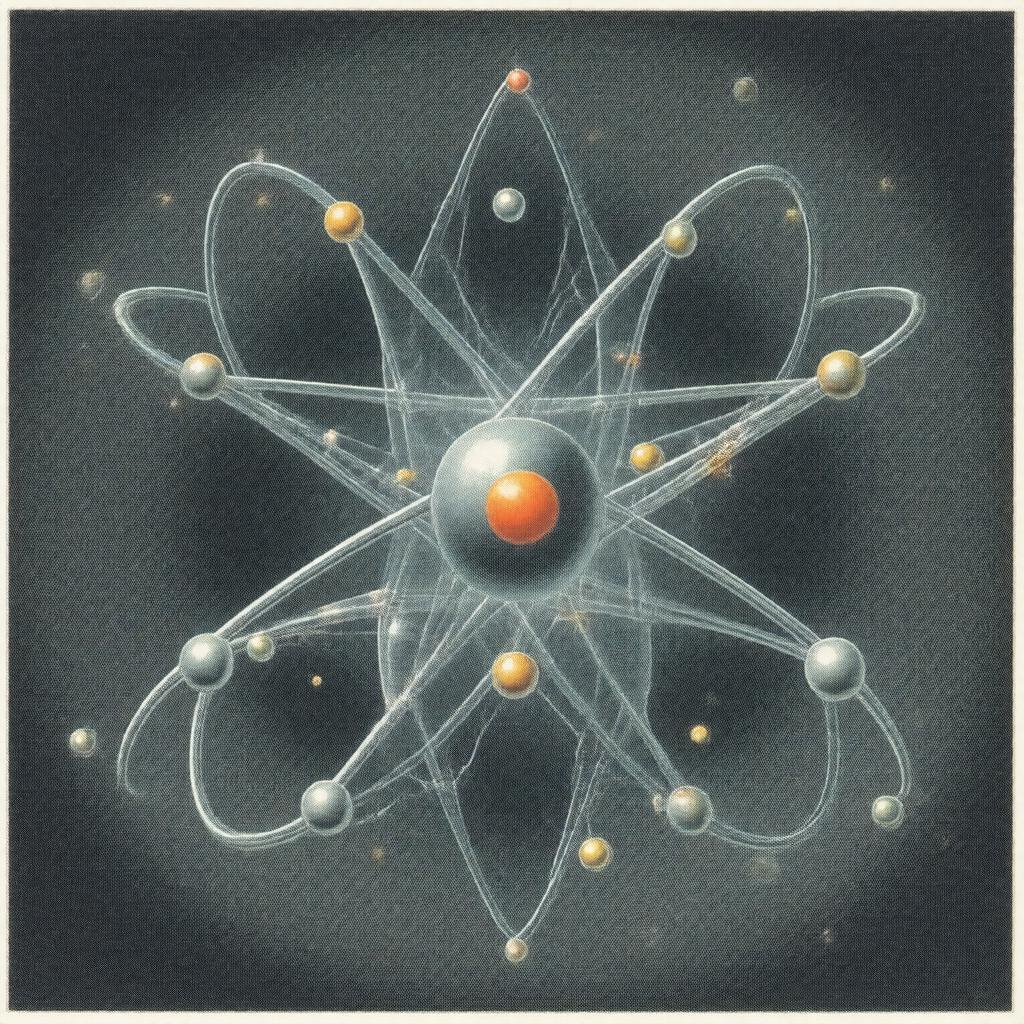
| Caption | Bohr model of the hydrogen atom |
| Field | Physics |
| Subdisciplines | Quantum mechanics; Spectroscopy; Laser physics; Plasma physics |
| Notable persons | Niels Bohr; Ernest Rutherford; J. J. Thomson; Max Planck; Erwin Schrödinger |
Atomic Physics Atomic Physics is the branch of physics that examines the properties and behavior of isolated atoms, their electronic structure, interactions, and spectra. It links foundational work by pioneers such as Niels Bohr, Ernest Rutherford, J. J. Thomson, Max Planck, and Erwin Schrödinger to contemporary research in precision measurement, quantum control, and atomic-based technologies. Through experimental and theoretical approaches developed in institutions like the Cavendish Laboratory, Bell Labs, MIT, and the Max Planck Society, Atomic Physics informs fields from metrology to astrophysics.
Overview and Scope
Atomic Physics addresses the electronic structure of atoms, bound and continuum states, and atom-light and atom-atom interactions. Core topics include electronic configuration, fine structure, hyperfine structure, isotope shifts, collision processes, and ionization dynamics investigated in laboratories such as Lawrence Berkeley National Laboratory, Rutherford Appleton Laboratory, and Los Alamos National Laboratory. Research often involves collaborations with observatories like Palomar Observatory and facilities such as the European Organization for Nuclear Research where atomic data support high-energy experiments. The scope overlaps with quantum optics, condensed matter in contexts like Bell Labs experiments, and plasma studies at centers like Princeton Plasma Physics Laboratory.
Atomic Structure and Models
Models of atomic structure evolved from the Thomson model to the Rutherford model, to the Bohr model and then to quantum treatments by Erwin Schrödinger and Werner Heisenberg. Quantum mechanical descriptions employ operators, wavefunctions, and the Pauli exclusion principle to explain electronic shells, subshells, and term symbols; experimental confirmations came from spectroscopic work by Angstrom, Fraunhofer, and Kirchhoff. Many-electron problems rely on approximations developed at institutions like Harvard University and Stanford University including Hartree–Fock, configuration interaction, and density functional approaches inspired by Walter Kohn and John Pople. Relativistic corrections use methods from Paul Dirac and Viktor Weisskopf; hyperfine interactions reference nuclear moments studied by Isidor Rabi and Enrico Fermi.
Spectroscopy and Transitions
Spectroscopy is central, linking observed lines to transitions predicted by selection rules derived by Arnold Sommerfeld, Max Born, and Wolfgang Pauli. Techniques include absorption, emission, fluorescence, and photoelectron spectroscopy developed in laboratories like Rutherford Laboratory and Columbia University. Precision frequency standards derive from atomic clocks based on transitions in Cesium-133, Rubidium, and optical standards using Strontium or Ytterbium ions; metrology is advanced at organizations such as the National Institute of Standards and Technology and Physikalisch-Technische Bundesanstalt. Astrophysical spectra from sources observed by the Hubble Space Telescope and the Very Large Telescope rely on lab atomic data by groups at Cambridge University and Caltech.
Experimental Methods and Techniques
Experimental methods include beam experiments pioneered by Ernest Rutherford and James Chadwick, laser cooling inspired by work at Bell Labs and MIT, magneto-optical traps developed by researchers at National Institute of Standards and Technology and University of Colorado Boulder, and ion trap techniques from Paul"" and Hans Dehmelt. Synchrotron radiation sources at SLAC National Accelerator Laboratory and European Synchrotron Radiation Facility enable photoabsorption studies; free-electron lasers at DESY and Lawrence Livermore National Laboratory permit ultrafast probing. Collisional and plasma diagnostics draw on apparatus used at Culham Centre for Fusion Energy and General Atomics; precision spectroscopy benefits from frequency combs pioneered by Theodor Hänsch and John Hall.
Applications and Technologies
Atomic Physics underpins atomic clocks used in the Global Positioning System and timing infrastructures at NIST, quantum sensors applied in geodesy and navigation by groups at Oxford University and Imperial College London, and quantum information processing using trapped ions at University of Innsbruck and University of Maryland. Laser technology developed through collaborations involving Bell Labs and Corning Incorporated enables applications from telecommunications to lidar used by aerospace firms like Boeing and Airbus. Isotope separation and atomic-beam techniques have roles in nuclear medicine at centers such as Mayo Clinic and Cleveland Clinic; spectroscopy aids material analysis in industry partners like Sandia National Laboratories and General Electric.
Historical Development and Key Figures
The field’s history traces major milestones: J. J. Thomson’s electron discovery, Ernest Rutherford’s scattering experiments, Niels Bohr’s quantized orbits, Max Planck’s quantum hypothesis, and the formulation of quantum mechanics by Erwin Schrödinger and Werner Heisenberg. Experimental advances include the maser and laser developed by Charles Townes, Theodore Maiman, and Arthur Schawlow, and atomic beam magnetic resonance by Isidor Rabi. Nobel laureates influential in the field include Wolfgang Pauli, Paul Dirac, Richard Feynman, Leon Cooper, and John Bardeen whose broader work impacted atomic theory. Institutions such as the Cavendish Laboratory, Institut d'Optique, National Institute of Standards and Technology, and Max Planck Institute for Quantum Optics served as hubs for key developments; conferences like the Solvay Conference and the Enrico Fermi Summer School fostered theoretical exchange.